
Have you ever struggled to find the information you need in a leaflet? Perhaps you’ve even given up on using it because you couldn’t fold it back up correctly. A better solution would be to access the instructions for your medication on your smartphone. This would save you time and frustration. If you are familiar with these thoughts or are interested in the topic from a regulatory perspective, you have come to the right place. Our article is about electronic product information for medicines or ePI for short.
In January 2020, the European Medicines Agency (EMA), the European Medicines Agency (HMA), and the European Commission came together. They defined principles that should apply to the development of a digital package leaflet. Outside of Europe, there are several pilot projects on this topic with exciting approaches, too.
What is electronic product information (ePI)?
In Europe and most other countries, the provision of a package leaflet is required by law. It informs doctors, pharmacists, and patients about the safe use of a medicine. The electronic version of the product information pursues the same goal. In addition to the instructions for use, it contains a summary of product characteristics (SmPC) and the labeling requirements for the pharmaceutical packaging.
An ePI is usually a file in XML, JSON, or RDF format. Compared to other digital standards such as MS Word or PDF, data can be easily extracted from this format without losing its external structure. This simplifies the electronic exchange of information between authorities and manufacturers via web portals or digital platforms and enables digital post-processing.
What are the advantages?
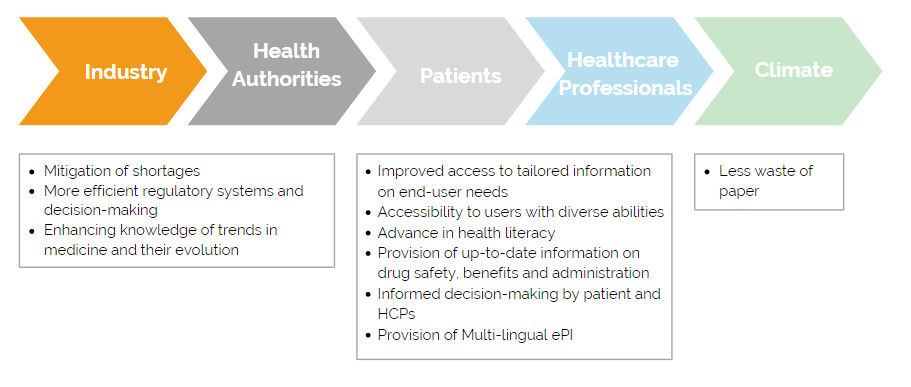
The coronavirus crisis has shown how important it is to work together digitally and make decisions in a short time. That also applies to the exchange of information between industry and authorities. Manufacturers can use ePIs to regularly forward reports on current therapeutic procedures, diseases, and their causes to the authorities. The latter could be informed more quickly by the industry about drug shortages. This way they can take more efficient measures, such as the redistribution of drugs to other EU member states.
With the aid of ePIs, patients and healthcare professionals will always have access to up-to-date information on the respective medicine. Regulatory changes to product information would not have to wait for the next printing of a new package leaflet. Furthermore, patients could read the package leaflet in their preferred language, regardless of the country they purchased the medicine in. Depending on the situation, a paper version of the package leaflet could be completely avoided in the future.
What are the challenges?
In addition to the obvious advantages, the development of ePIs brings with it some challenges. An open dialogue between all parties is important for coordinating such a big project. For example, when deciding on a standardized ePI format, sufficient funding and embedding the ePIs in existing national or international telematics structures.
One issue frequently discussed internationally and handled differently depending on the country is how patients are given access to the digital patient information leaflet. India proposes to make the product information available on the manufacturer’s website. In contrast, Taiwan plans to centralize data management through the TFDA. If the website link is embedded in a QR code, it can be scanned with any smartphone but takes up additional space on the pharmaceutical packaging. Therefore, many pharmaceutical representatives propose integrating the website link into the GS1 data matrix and developing a scanner app as part of an international project. In the United Arab Emirates, it has been decided to avoid the discussion altogether and introduce both codes as mandatory. For all other countries, GS1 recommends contacting local authorities and GS1 representatives to find out more and, if necessary, to influence the emerging regulations.
What is the EU planning?
On December 11, 2025, representatives of the European Parliament, the Council, and the Commission agreed on far-reaching changes to the pharmaceutical legislation. Among the points discussed was the electronic package insert, which is set to become mandatory for all medicines. On March 18, 2026, the European Union’s responsible Subcommittee on Public Health (SANT) also approved the revision. The content of the new pharmaceutical legislation must now be translated into all languages of the European Union. Afterwards it can be confirmed by the Parliament and the Council. Until final confirmation, this process is expected to continue until fall 2026.
According to the new directive, marketing authorization holders will be required to provide patient information leaflet for their medicines in both printed and digital formats in the future. Individual member states will also have the option to waive the requirement for a printed version for selected medicines or groups of medicines. However, this must be preceded by a consultation with patients. If necessary, the option for a printed version must still be available.
Should the new pharmaceutical law be confirmed as planned in the fall of 2026, there will be a transition period of 24 months. EU member states would then have until mid-2028 to implement these changes.
[Disclaimer]
The information provided here is only one possible interpretation of the regulations. These are also subject to constant change, so the information in this article may be incomplete or no longer up to date. The above article is expressly not intended as legal advice. Please consult the official documents before making any business decisions. (Information status: April 2026)